Record-Keeping in Retail Pharmacy Businesses
Robust record-keeping in pharmacies is a legal requirement that supports patient care, evidence-based practice, professional accountability and effective management of the medicines supply chain.
Overview
Accurate, complete and compliant record-keeping is a cornerstone of safe and effective pharmacy practice and patient care. Pharmacy records play a crucial role in safeguarding patients, supporting clinical and professional decision-making and ensuring the safe supply of medicinal products. They also demonstrate compliance with pharmacy and medicines legislation and with professional standards and guidance.
Recent changes to enable electronic record-keeping in pharmacies
Recent legislative amendments have been introduced to enable pharmacies to keep the following pharmacy records electronically, provided that the pharmacy computer software system in use meets certain specificed minimum requirements:
- Duty Register
- Prescription Register/Daily Audit
- Controlled Drugs Register
- Prescriptions transferred through Healthmail and the High-Tech Hub.
Changes to legislation to allow for electronic record-keeping reduce administrative burden for pharmacists and are a very positive step forward to modernise record-keeping requirements in community pharmacies. These changes are also an important interim step towards the development of the national ePrescribing solution and wider digitisation in pharmacies and our health system.
The following statutory instruments were signed into law by the Minister for Health in April 2026 to enable electronic record-keeping and they are effective from 30 June 2026:
- Medicinal Products (Prescription and Control of Supply) (Amendment) Regulations 2026 (S.I. No. 138/2026)
- Misuse of Drugs (Amendment) (No. 2) Regulations 2026 (S.I. No. 139/2026)
- Regulation of Retail Pharmacy Businesses (Amendment) Regulations 2026 (S.I. No. 140/2026)
The option to continue keeping these records in paper-based format is retained in the legislation. In circumstances where the pharmacy computer software system does not meet the minimum required standard, pharmacies must continue to keep these records in paper-based format.
New additional record keeping requirements
In addition, the following new record keeping requirements will also come into operation on 30 June 2026:
- Pharmacists will be required to include their registration number also on paper-based prescriptions as part of endorsing requirements.
- All pharmacists (including superintendents and pharmacy owners) working in a pharmacy will be required to record their details in the daily duty register.
Including these changes now, provides for greater transparency and traceability of the pharmacist involved in the dispensing of a prescription and prepares for the rollout in the near future of national digital health systems.
Next steps to develop pharmacy software and enable implementation of electronic record-keeping
These changes are effective from 30 June 2026. Pharmacies will only be able to keep these records electronically when the pharmacy owner can verify that their pharmacy computer software meets the minimum requirements as specified in legislation.
The next steps to enable the development of pharmacy software and prepare for implementation are being advanced by the various pharmacy stakeholders on the Department of Health Electronic Record-Keeping Implementation Group. This multi-stakeholder group has been established to progress legislation and implementation steps that enable electronic record-keeping and reduce administrative burden in pharmacies. We will provide further information on implementation when available.
Further information on the technology requirements and endorsing requirements can be found in our revised draft PSI Guidelines on Record-Keeping in a Retail Pharmacy Business. These were published recently for public consultation feedback - see the section below. In the meantime, you'll find information in the FAQs at the end of this page.
PSI Guidelines on Record-Keeping in a Retail Pharmacy Business
We have updated our record-keeping Guidelines to reflect the changes to the legislation. We published the draft guidelines for public consultation from 21 April to 19 May 2026 and are currently reviewing the feedback received. The updated guidelines will be published following the PSI Council's consideration of the consultation report and the guidelines at their June meeting.
In line with recent guidance and guidelines that we have issued, these Guidelines have undergone significant review and update and have been reformatted as principles-based Guidelines which are intended to be supportive, enabling, and explain and facilitate compliance with the legislative requirements for record-keeping. The Guidelines comprise five guiding principles designed to support consistent, safe, compliant and high-quality record-keeping in pharmacies.
-
What changes to record-keeping will come into operation on 30 June 2026, and why?
From 30 June 2026, the following changes to record-keeping will come into operation in retail pharmacy businesses:
a. Recent legislative amendments will enable pharmacies to keep the following records in electronic form, where they choose to do so, provided that the pharmacy computer software system in use meets certain specified minimum requirements:
- Duty Register
- Prescription Register/Daily Audit
- Controlled Drugs Register
- Prescriptions transferred through Healthmail and the High Tech Hub.
b. Pharmacists will be required to include their registration number also on paper-based prescriptions as part of endorsing requirements.
c. All pharmacists (including superintendents and pharmacy owners) working in pharmacies will be obliged to record their details in the duty register.
Including these changes now, provides for greater transparency and traceability of the pharmacist involved in the dispensing of a prescription and prepares for the rollout in the near future of national digital health systems.
Pharmacies will only be able to keep these records electronically when the pharmacy owner can verify that their pharmacy computer software meets the minimum requirements as specified in legislation.
The option to continue keeping these records in paper-based format is retained in the legislation. In circumstances where the pharmacy computer software system does not meet the minimum required standard, pharmacies must continue to keep these records in paper-based format.
This follows commitments made by the Department of Health under the Community Pharmacy Agreement 2025 to prioritise actions on regulatory, administrative and process reforms aimed at reducing administrative burden in community pharmacies.
The changes are outlined in detail in these FAQs and on the page above.
2. What legislation has been amended?
Changes have been made to the following regulations to facilitate the move to electronic record-keeping:
- Medicinal Products (Prescription and Control of Supply) Regulations 2003 (S.I. 540 of 2003) (as amended)
- Misuse of Drugs Regulations 2017 (S.I. No. 173 of 2017) (as amended)
- Regulation of Retail Pharmacy Businesses Regulations 2008 (S.I. No. 488 of 2008) (as amended).
The following statutory instruments were signed into law by the Minister for Health on 9 April to enable these changes to record-keeping and will come into operation on 30 June 2026:
- Medicinal Products (Prescription and Control of Supply) (Amendment) Regulations 2026 (S.I. No. 138/2026)
- Misuse of Drugs (Amendment) (No. 2) Regulations 2026 (S.I. No. 139/2026)
- Regulation of Retail Pharmacy Businesses (Amendment) Regulations 2026 (S.I. No. 140/2026)
All legislation can be accessed in full through www.irishstatutebook.ie.
3. Do I have to transition to keeping these records electronically?
No. Pharmacies are not required to transition to electronic record-keeping. The option to continue keeping these records in paper-based format is retained in the legislation.
4. When will the changes be implemented?
These changes are effective from 30 June 2026. Pharmacies will only be able to keep these records electronically when the pharmacy owner can verify that their pharmacy computer software meets the minimum requirements as specified in legislation.
For further information, see the Technology Requirements section below and the PSI Guidelines on Record-Keeping in a Retail Pharmacy Business.
The Department of Health has established a multi-stakeholder group with the various pharmacy stakeholders, including the PSI, IPU and HSE. This group has worked together to progress the legislation and will coordinate the required updates to pharmacy software systems and prepare for the implementation of electronic record-keeping in pharmacies. This work is ongoing, and we will provide further information on implementation when available.
5. What is the role of the Department of Health Electronic Record-Keeping Implementation Group?
In November 2025, the Department of Health established a multi-stakeholder Electronic Record-Keeping Implementation Group. The role of the group is to progress legislative change aimed at modernising prescription management, supporting the adoption of electronic record-keeping practices to reduce administrative burden in pharmacies and enabling a more streamlined and digitally supported pharmacy workflow.
The Group is chaired by the Department of Health and includes representation from:
- PSI
- Irish Pharmacy Union (IPU)
- Health Service Executive Primary Care Reimbursement Service (HSE-PCRS)
- Health Service Executive National Quality and Patient Safety Directorate (HSE-QPS)
- HSE Access and Integration Drug Management Programme (HSE -AIDMP)
- HSE ePharmacy
- Hospital Pharmacists Association of Ireland (HPAI)(Retail Pharmacy Business Representation)
- Irish Platform for Patient Organisations, Science and Industry (IPPOSI)
The PSI’s role on the group includes leading on the development of regulatory guidelines and supports, where appropriate, to assist in the safe implementation of electronic record-keeping in pharmacies.
This group has worked together to progress the legislation and will coordinate the required updates to pharmacy software systems and prepare for implementation of electronic record-keeping in pharmacies. This work is ongoing and we will provide further information on implementation when available.
6. Can hospital pharmacies keep pharmacy records electronically?
The revised legislation only applies to registered retail pharmacy businesses:
-
What requirements does my pharmacy computer software need to meet in order to keep these records electronically?
In order to keep a controlled drugs register, prescription register/daily audit or duty register electronically, the pharmacy owner will be required to verify that their pharmacy computer software meets the requirements specified in legislation for each type of register.
Controlled Drugs Register
- The entries in the register are subject to user access controls capable of restricting the functions that may be used.
- Every correction to an entry in the register is capable of being traced by identifying the original entry, the correction to the original entry, the identity of the person who made the correction and the date of the correction.
- Every entry in the register is capable of being searched, sorted and reproduced by:
- The date on which the supply was received or the transaction was effected
- The name and address of the person from whom the product was obtained or to whom the product was supplied
- The authority of the person supplied to be in possession, where applicable (e.g. prescription reference)
- The product
Prescription Register/Daily Audit
- The entries in the register are subject to user access controls capable of restricting the functions that may be used.
- Every alteration to an entry in the register is capable of being traced by identifying the original entry, the alteration to the original entry, the identity of the person who made the alteration and the date of the alteration.
- Every entry in the register is capable of being searched, sorted and reproduced by:
- The date on which the medicinal product was supplied
- The name of the medicinal product
- The strength of the medicinal product, where applicable
- The name of the person for whom the medicinal product was prescribed or to whom the product was supplied
- The name of the prescriber, where applicable
Duty Register
- The record is subject to user access controls capable of restricting the functions that may be used.
- Every alteration to an entry in the record is capable of being traced by identifying the original entry, the alteration to the original entry, the identity of the person who made the alteration and the date of the alteration.
8. How will I know that my pharmacy computer software system meets the requirements specified in the legislation?
As outlined above in question 4, the next steps to enable the development of pharmacy software and prepare for implementation are being advanced by the various pharmacy stakeholders on the Department of Health Electronic Record-Keeping Implementation Group. Further information will be shared once available and in advance of 30 June 2026.
Please note, pharmacies will not be able to keep these records electronically until the pharmacy owner can verify that their pharmacy computer software meets the requirements specified in legislation.
Further information on how pharmacy owners will be able to verify that their pharmacy computer software meets the requirements specified in legislation for electronic record-keeping will be shared once available.
-
What changes have been made to the endorsing requirements for paper-based prescriptions?
Changes have been introduced to the legislation which require pharmacists, from 30 June 2026, to include their registration number on paper-based prescriptions as part of endorsing requirements. This provides for greater transparency and traceability of the pharmacist involved in the dispensing of a prescription and the supply of a medicine.
This change is being introduced now to improve record-keeping and to prepare for the rollout in the near future of national digital health systems under development, such as the National Shared Care Record (NSCR) and the National ePrescribing Service (NePS).
All health practitioners, including registered pharmacists, will need to be identifiable in these new electronic systems.
-
What details must I record on a fully dispensed paper-based prescription?
From 30 June 2026, for all fully dispensed paper-based prescriptions, the following details must be recorded on the prescription:
- Date
- Registration number of the pharmacist who dispensed the prescription
- The word “dispensed” written or printed prominently
-
What details must I record on a partially dispensed paper-based prescription?
From 30 June 2026, for all partially dispensed paper-based prescriptions, the following details must be endorsed on the prescription:
- Date
- Registration number of the pharmacist who dispensed the prescription
- Name and address of the pharmacy
- Quantity dispensed
-
Do I still need to print Healthmail prescriptions?
Pharmacy owners are required to ensure that all prescriptions transmitted by the National Electronic Prescription Transfer System, i.e. Healthmail and the High-Tech Hub, are preserved as transmitted and kept readily available for inspection at the pharmacy.
In addition, if a pharmacy is continuing to maintain pharmacy records in paper-based format, they are required to print a copy of the Healthmail and/or High-Tech Hub prescription and treat the printed copy as an original prescription for the purposes of record-keeping, as per the current requirements.
From 30 June 2026, if a pharmacy is retaining Healthmail and/or High-Tech Hub prescriptions electronically, they must preserve an electronic copy of the prescription within the Patient Medication Record (PMR) (see questions 13 and 14 below).
-
What must I do if I want to retain Healthmail and High-Tech Hub prescriptions electronically?
All prescriptions transmitted via Healthmail and/or the High-Tech Hub must be preserved as transmitted.
Where a pharmacy is retaining records electronically, an electronic copy of the prescription must be preserved within the Patient Medication Record (PMR) and treated as an original prescription for the purposes of record-keeping.
The electronic copy of the prescription within the PMR must be preserved in such a manner to enable the ready identification of both the prescription used to authorise the dispensing of the medicinal product(s) and the medicinal product(s) previously dispensed under that prescription, where applicable.
14. What information must I record in the Patient Medication Record (PMR) when keeping Healthmail and High-Tech prescriptions electronically?
The table below outlines the information that must be recorded in the PMR when preserving Healthmail and High-Tech Hub prescriptions electronically.
| CD2&3 | CD4 Part 1 – Fully Dispensed | CD4 Part 1 – Partially Dispensed | Non-CD – Fully Dispensed | Non-CD Partially Dispensed | |
|---|---|---|---|---|---|
| Date of dispensing | ✓ | ✓ | ✓ | ✓ | ✓ |
| Registration number of pharmacist | ✓ | ✓ | ✓ | ✓ | ✓ |
| Quantity dispensed | X | X | ✓ | X | ✓ |
| Preserve the prescription at the pharmacy premises* |
✓ (Preserve the prescription as transmitted and an electronic copy of the prescription within the Patient Medication Record (PMR)) |
✓ (Preserve the prescription as transmitted and an electronic copy of the prescription within the Patient Medication Record (PMR)) |
✓ (Preserve the prescription as transmitted and an electronic copy of the prescription within the Patient Medication Record (PMR)) |
✓ (Preserve the prescription as transmitted and an electronic copy of the prescription within the Patient Medication Record (PMR)) |
✓ (Preserve the prescription as transmitted and an electronic copy of the prescription within the Patient Medication Record (PMR)) |
| * The electronic copy of the prescription within the PMR must be preserved in such a manner which shall enable the ready identification of the prescription used to authorise the dispensing of the medicinal product(s) and the medicinal product(s) previously dispensed under that prescription, where applicable. | |||||
-
Can I keep paper-based prescriptions received electronically?
No. Any prescriptions received in paper-based format must continue to be kept as per paper-based record-keeping requirements. Only prescriptions received via the National Electronic Prescription Transfer System, i.e. Healthmail and the High-Tech Hub, can be maintained electronically and kept as per electronic record-keeping requirements.
-
What do I need to do to keep the daily audit/prescription register electronically?
To keep the daily audit/prescription register electronically, the pharmacy owner must verify that the computer software in use for the retention of the records meets the following requirements:
- The entries in the register are subject to user access controls capable of restricting the functions that may be used.
- Every alteration to an entry in the register is capable of being traced by identifying the original entry, the alteration to the original entry, the identity of the person who made the alteration and the date of the alteration.
- Every entry in the register is capable of being searched, sorted and reproduced by:
- The date on which the medicinal product was supplied
- The name of the medicinal product
- The strength of the medicinal product, where applicable
- The name of the person for whom the medicinal product was prescribed or to whom the product was supplied
- The name of the prescriber, where applicable
The Department of Health multi-stakeholder group will coordinate the required updates to pharmacy software systems and prepare for the implementation of electronic record-keeping in pharmacies. This work is ongoing, and we will provide further information on implementation when available.
Additionally, pharmacists and those in statutory governance roles must consider the five guiding principles and applicable indicators outlined in the PSI Guidelines on Record-Keeping in a Retail Pharmacy Business when implementing an electronic daily audit/prescription register to comply with the requirements set out in legislation and in the PSI Guidelines. Appendix 1 of the PSI Guidelines on Record-Keeping outlines the record-keeping requirements for the daily audit/prescription register.
-
Can I still print a paper-based daily audit/prescription register on a daily basis, if I don’t want to move to electronic retention?
Yes. The option to continue keeping the daily audit/prescription register in paper-based format is retained in the legislation.
-
What do I need to do to keep the controlled drugs register electronically?
To keep the controlled drugs register electronically, the pharmacy owner must verify that the computer software in use for the retention of the records meets the following requirements:
- The entries in the register are subject to user access controls capable of restricting the functions that may be used.
- Every correction to an entry in the register is capable of being traced by identifying the original entry, the correction to the original entry, the identity of the person who made the correction and the date of the correction.
- Every entry in the register is capable of being searched, sorted and reproduced by:
- The date on which the supply was received or the transaction was effected
- The name and address of the person from whom the product was obtained or to whom the product was supplied
- The authority of the person supplied to be in possession, where applicable (e.g. prescription reference)
- The product
The Department of Health multi-stakeholder group will coordinate the required updates to pharmacy software systems and prepare for the implementation of electronic record-keeping in pharmacies. This work is ongoing, and we will provide further information on implementation when available.
Additionally, pharmacists and those in statutory governance roles must consider the five guiding principles and applicable indicators outlined in the PSI Guidelines on Record-Keeping in a Retail Pharmacy Business when implementing an electronic controlled drugs register to comply with the requirements set out in legislation and in the PSI Guidelines. Appendix 2 of the PSI Guidelines on Record-Keeping outlines the record-keeping requirements for the controlled drugs register.
-
Can I keep Schedule 2, 3 or 4 controlled drug destruction records electronically?
Yes. The register may be an electronic register which is based on a computerised system equivalent to the register. Further information is outlined in question 18 above. In addition, the record must include the following details:
- Date of destruction
- Name of the controlled drug
- Quantity destroyed
- Digitally signed by the authorised person in whose presence the controlled drug is destroyed.
-
Can I still keep a paper-based controlled drugs register if I don’t want to move to electronic retention?
Yes. The option to continue keeping the controlled drugs register in paper-based format is retained in the legislation.
-
What changes have been made to the details that must be recorded in the duty register?
From 30 June 2026, all registered pharmacists on duty must record their details in the duty register, whether it is being maintained as a paper-based or electronic record. The pharmacy owner and superintendent pharmacist must ensure that an ongoing, contemporaneous and retrievable record of all registered pharmacists on duty is maintained and is available at the pharmacy premises.
-
What do I need to do to keep the duty register electronically?
To keep the duty register electronically, the pharmacy owner must verify that the computer software in use for the retention of the records meets the following requirements:
- The record is subject to user access controls capable of restricting functions that may be used.
- Every alteration to an entry in the record is capable of being traced by identifying the original entry, the alteration to the original entry, the identity of the person who made the alteration and the date of the alteration.
Additionally, pharmacists and those in statutory governance roles must consider the five guiding principles and applicable indicators outlined in the PSI Guidelines on Record-Keeping in a Retail Pharmacy Business when implementing an electronic duty register to comply with the requirements set out in legislation and in the PSI Guidelines.
-
Can I still keep a paper-based duty register if I don’t want to move to electronic retention?
Yes. The option to continue keeping the duty register in paper-based format is retained in the legislation.
-
How long do I have to keep the various pharmacy records in paper-based or electronic format?
| Record | Retention Period | Where |
|---|---|---|
| Prescriptions (both paper and electronically transmitted) | 2 years | Available* at the pharmacy premises |
| Orders and invoices, or a copy of these, relating to the supply of medicinal products to a registered medical practitioner, registered dentist, or registered veterinary surgeon for administration to a patient in the course of their professional practice | 2 years | Available at the pharmacy premises |
| Prescription Register/Daily Audit | 2 years from date of the last entry made in the register | Available at the pharmacy premises |
| Register of Controlled Drugs | 2 years from date of the last entry made in the register | Available at the pharmacy premises |
| Orders, prescriptions or requisitions against which a controlled drug is supplied | 2 years | Available at the pharmacy premises |
| Paper or electronic copies of Schedule 4 Part 1 controlled drug repeatable prescriptions, which are dispensed in part and any endorsements made | 2 years | Available at the pharmacy premises |
| Invoices, or other like record, issued in respect of all Schedule 3 and Schedule 4 Part 1 controlled drugs obtained or supplied from the pharmacy | 2 years | Recommended to be available at the pharmacy premises, but not specified in legislation |
| Invoices, or other like record, issued in respect of all Schedule 5 controlled drugs obtained by the pharmacy | 2 years | Recommended to be available at the pharmacy premises, but not specified in legislation |
| Records in relation to sale or supply of exempt medicinal products | 5 years |
Available at the pharmacy premises
|
| An ongoing, contemporaneous and retrievable record of all registered pharmacists on duty i.e. the Duty Register | 2 years from date of last entry made in the register | Available at the pharmacy premises |
| Records to show the safe operation of the pharmacy, e.g. fridge and storage room temperature records, invoices for medicinal products not listed above | Recommended minimum 2 years, but not specified in legislation | Recommended to be available at the pharmacy premises, but not specified in legislation |
| *Available means that both paper and electronic formats are acceptable | ||
-
Along with the pharmacy computer software system requirements, is there anything else I need to have in place to transition to electronic record-keeping?
We have updated our record-keeping guidelines to reflect the changes to the legislation. These are currently available in draft. The guidelines were published for public consultation from 21 April to 19 May 2026 and we are currently reviewing the feedback received. The updated guidelines will be published following the PSI Council's consideration of the consultation report and the guidelines at their June meeting.
The PSI Guidelines on Record-Keeping in a Retail Pharmacy Business (draft) set out five guiding principles (see figure 1 below) to support pharmacists and those in statutory governance roles in implementing the record management system best suited to their pharmacy. Each principle is underpinned by concise, outcome-focused indicators that offer guidance on how adherence to each principle can be demonstrated. Pharmacists and those in statutory governance roles must have regard to the relevant principles and indicators when implementing a record management system appropriate to their retail pharmacy business, to comply with legislative requirements and the PSI Guidelines.
The Guidelines are intended to be supportive and enabling, outlining the legislative requirements while allowing flexibility in how pharmacies implement their chosen record management system.
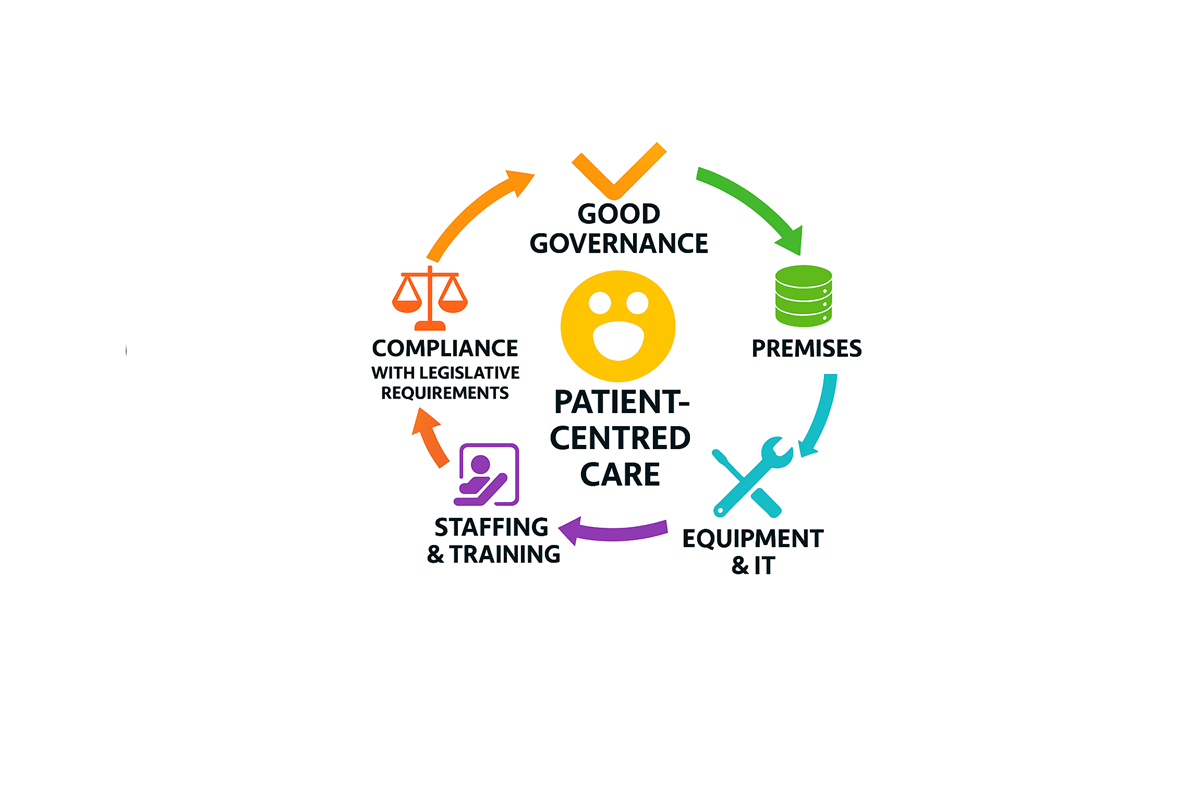
In addition, the Department of Health Electronic Record-Keeping Implementation Group is also considering other supports, such as operational guidance and training for pharmacists and their teams that will be needed to support the safe transition to electronic record-keeping. Further information on these additional supports will be shared in due course, once available.

